Your Cart is Empty
Free Shipping on ALL alarms!
And on all orders of $35 or more *Continental US only
Free Shipping on ALL alarms!
And on all orders of $35 or more *Continental US only
J-FMKN95
KN95 face masks work by using a 4 layer filtration system to filter out airborne particles such as harmful pollutants, dust, smoke, fiberglass, wood, and other allergens allowing you to breathe easier, and protect your lungs. Each mask is disposable, and by wearing a new mask each time you are going to come into contact with airborne allergens or pollutants helps ensure maximum filtration to protect your respiratory system. To ensure proper use, the mask should fit snugly to the contour of your face. You can adjust the fit of the mask using the adjustable nose bridge, and secure the mask comfortably behind your ears with the soft elastic ear loops designed to eliminate pressure to the outer ear. Masks should fit most adults and children, and can be used year round when visiting public areas such as grocery stores, pharmacies, parks, and post offices.
Note: These masks are not NIOSH certified.. These masks are intended for use by non-medical personnel and no claim is being made about their effectiveness against specific pathogens. U.S. health care providers should consult the FDA guidance at this link prior to use. The manufacturer of this mask is an establishment registered with the FDA. Testing by the National Personal Protective Technology Laboratory indicates a maximum filter efficiency for this mask at 90.04%
Frequently Asked Questions
Q: What is a KN95 mask and how does it compare to an N95 respirator mask.
A: N95 masks are manufactured to meet a certain U.S. test standard defined by NIOSH (National Institute for Occupational Safety and Health). The 3M company is probably the best known for their N95 respirators in the U.S. KN95 masks are manufactured in China and certified against a similar standard.
Q: How do I know if the KN95 mask I buy meets the certification standards.
A: Our KN95 masks are sourced from reputable manufacturers in China that are registered with the U.S. FDA.
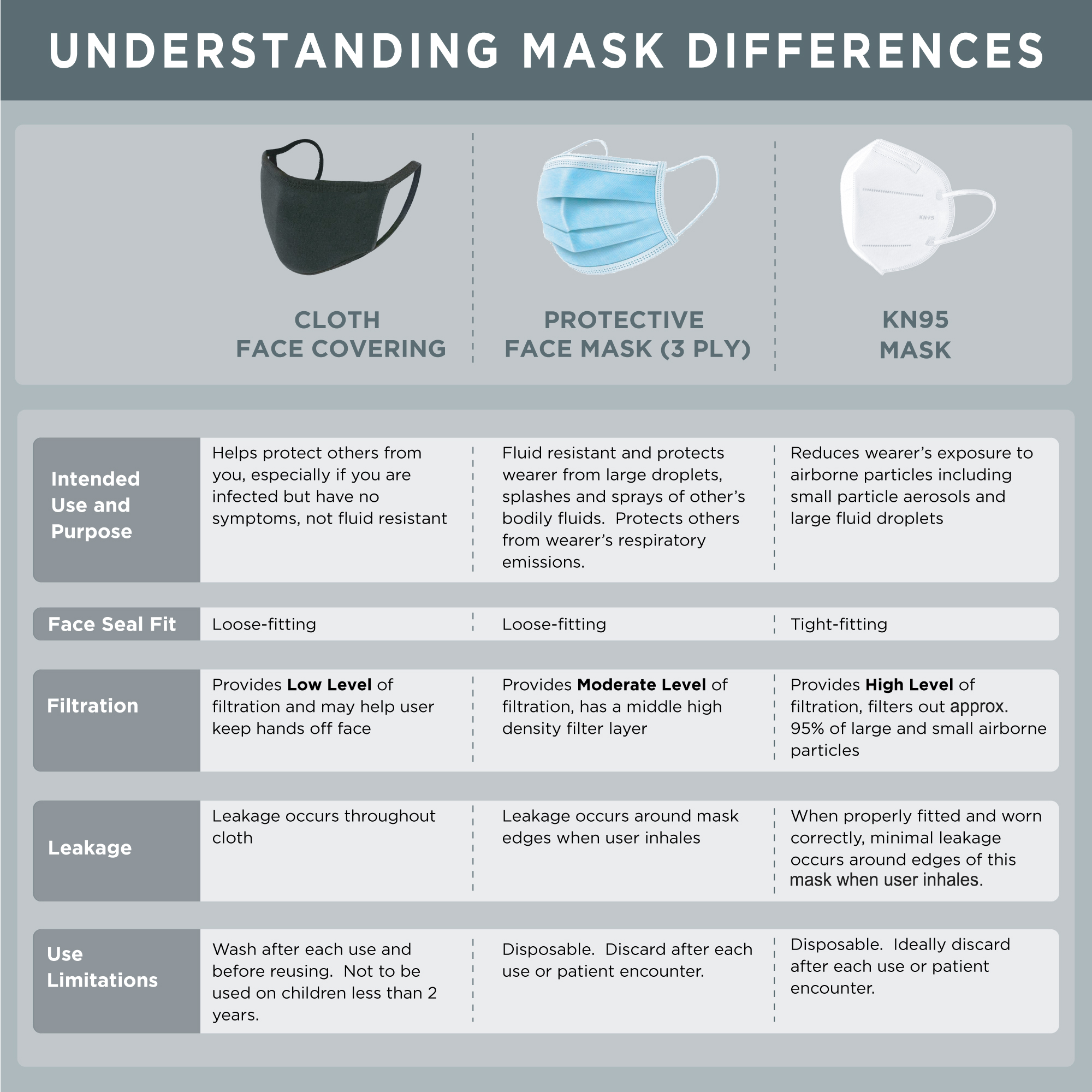
KN95 face masks work by using a 4 layer filtration system to filter out airborne particles such as harmful pollutants, dust, smoke, fiberglass, wood, and other allergens allowing you to breathe easier, and protect your lungs. Each mask is disposable, and by wearing a new mask each time you are going to come into contact with airborne allergens or pollutants helps ensure maximum filtration to protect your respiratory system. To ensure proper use, the mask should fit snugly to the contour of your face. You can adjust the fit of the mask using the adjustable nose bridge, and secure the mask comfortably behind your ears with the soft elastic ear loops designed to eliminate pressure to the outer ear. Masks should fit most adults and children, and can be used year round when visiting public areas such as grocery stores, pharmacies, parks, and post offices.
Note: These masks are not NIOSH certified.. These masks are intended for use by non-medical personnel and no claim is being made about their effectiveness against specific pathogens. U.S. health care providers should consult the FDA guidance at this link prior to use. The manufacturer of this mask is an establishment registered with the FDA. Testing by the National Personal Protective Technology Laboratory indicates a maximum filter efficiency for this mask at 90.04%
Frequently Asked Questions
Q: What is a KN95 mask and how does it compare to an N95 respirator mask.
A: N95 masks are manufactured to meet a certain U.S. test standard defined by NIOSH (National Institute for Occupational Safety and Health). The 3M company is probably the best known for their N95 respirators in the U.S. KN95 masks are manufactured in China and certified against a similar standard.
Q: How do I know if the KN95 mask I buy meets the certification standards.
A: Our KN95 masks are sourced from reputable manufacturers in China that are registered with the U.S. FDA.

| MANUFACTURER ITEM CODE | SANAI MEDICAL TECHNOLOGY CO., LTD. 210-3 Yuejin Road, Xibin Town, Jinjiang City Quanzhou Fujian, CN 362221 Registration Number: 3016730678 FEI Number*: 3016730678 Status: Active Date Of Registration Status: 2020 |